- #Chantix free 30 day trial 2018 Patch#
- #Chantix free 30 day trial 2018 registration#
- #Chantix free 30 day trial 2018 trial#
- #Chantix free 30 day trial 2018 plus#
#Chantix free 30 day trial 2018 trial#
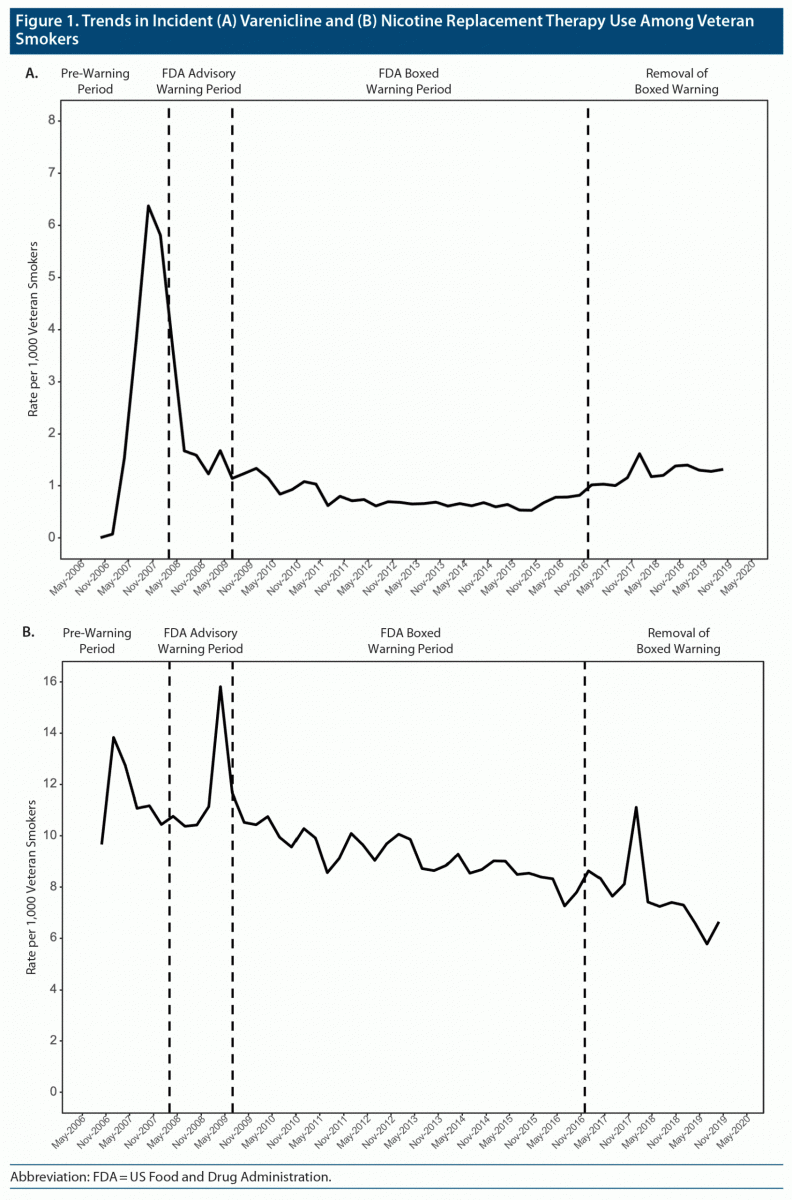
15 Subsequently, a number of publications have reached mixed conclusions regarding the safety of varenicline, including several meta-analyses, 11, 16 - 18 a retrospective cohort study, 19 a clinical trial among smokers with acute coronary syndrome, 20 and most recently, an observational study using pharmacy and health record administrative databases. 14 However, in 2011, the US Food and Drug Administration (FDA) mandated strengthened product warnings addressing the possibility of an increased CV event risk in smokers with established CVD. 13 Early clinical trials of varenicline, including a study of smokers with CVD, found rates of CV events (including MIs and strokes) to be low and not significantly higher than for placebo. However, clinical trials of bupropion in smokers with CVD have not identified an increased incidence of CV AEs. 9 - 11 Bupropion hydrochloride can increase blood pressure, 12 and the package label includes precautions about hypertension.
#Chantix free 30 day trial 2018 Patch#
Initial concerns regarding the risk of MI if a person smoked while wearing a nicotine patch 9 were dispelled, and nicotine replacement therapy (NRT) is now recognized as a safe treatment for smokers with CV disease (CVD). 5 - 8ĭespite the proven efficacy of smoking cessation medications, many clinicians have been hesitant to prescribe them because of concerns regarding adverse events (AEs), including CV safety. The US Public Health Service Clinical Practice Guideline for Smoking Cessation, as well as guidelines from other countries, recommend smoking cessation pharmacotherapy for all smokers making a quit attempt. 1 - 4 Quitting is the single most important step a cigarette smoker can take to protect and enhance cardiovascular (CV) health.
#Chantix free 30 day trial 2018 registration#
Trial Registration Identifier: NCT01574703Ĭigarette smoking is associated with an increased risk of myocardial infarction (MI), stroke, peripheral vascular disease, atrial fibrillation, sudden death, worsening heart failure, and increased rates of thrombosis following coronary revascularization. The findings of EAGLES and its extension trial provide further evidence that smoking cessation medications do not increase the risk of serious cardiovascular events in the general population of smokers.

There was no significant difference in time to onset of MACE for either varenicline or bupropion treatment vs placebo (varenicline: hazard ratio, 0.29 95% CI, 0.05-1.68 and bupropion: hazard ratio, 0.50 95% CI, 0.10-2.50).Ĭonclusions and Relevance No evidence that the use of smoking cessation pharmacotherapies increased the risk of serious cardiovascular adverse events during or after treatment was observed. No significant treatment differences were observed in time to cardiovascular events, blood pressure, or heart rate.

The incidence of cardiovascular events during treatment and follow-up was low (<0.5% for MACE <0.8% for MACE+) and did not differ significantly by treatment. Results Of the 8058 participants, 3553 (44.1%) were male (mean age, 46.5 years). Main Outcomes and Measures The primary end point was the time to development of a major adverse cardiovascular event (MACE: cardiovascular death, nonfatal myocardial infarction, or nonfatal stroke) during treatment secondary end points were the occurrence of MACE and other pertinent cardiovascular events (MACE+: MACE or new-onset or worsening peripheral vascular disease requiring intervention, coronary revascularization, or hospitalization for unstable angina). Interventions Varenicline, 1 mg twice daily bupropion hydrochloride, 150 mg twice daily and nicotine replacement therapy, 21-mg/d patch with tapering.
#Chantix free 30 day trial 2018 plus#
Smokers, with or without established psychiatric diagnoses, who received at least 1 dose of study medication (n = 8058), as well as a subset of those who completed 12 weeks of treatment plus 12 weeks of follow up and agreed to be followed up for an additional 28 weeks (n = 4595), were included. Objective To compare the relative cardiovascular safety risk of smoking cessation treatments.ĭesign, Setting, and Participants A double-blind, randomized, triple-dummy, placebo- and active-controlled trial (Evaluating Adverse Events in a Global Smoking Cessation Study ) and its nontreatment extension trial was conducted at 140 multinational centers. Importance Quitting smoking is enhanced by the use of pharmacotherapies, but concerns have been raised regarding the cardiovascular safety of such medications.